Team expects, may be useful, could be used, prototype, are currently investigating and so on. Cool piece of technolgy, but no even mention when they'd expect that to be commercially available, if it's even possible to manufacture in commercial scale. Like many other new battery chemistries and technologies, it shows promise and makes a good headline, but at this point that's pretty much it.
Technology
This is a most excellent place for technology news and articles.
Our Rules
- Follow the lemmy.world rules.
- Only tech related news or articles.
- Be excellent to each other!
- Mod approved content bots can post up to 10 articles per day.
- Threads asking for personal tech support may be deleted.
- Politics threads may be removed.
- No memes allowed as posts, OK to post as comments.
- Only approved bots from the list below, this includes using AI responses and summaries. To ask if your bot can be added please contact a mod.
- Check for duplicates before posting, duplicates may be removed
- Accounts 7 days and younger will have their posts automatically removed.
Approved Bots
Eh, give em the clout they need to develop it further.
Well tbf this was a university lab which isn't focused on commercial production but just trying to prove their experiments
To be fair, commercial long-life nickel-iron batteries are already being sold for grid storage. The main reason they aren't used more widely is they cost more up front.
That's ok, because they still cost less than alternatives over the full life span of the battery.
The risk is that the higher purchase cost required will likely be wasted as new battery tech surpasses it long before its life is over.
So for now, it's all about weighing opportunity cost, tech lock-in, and early obsolescence
We've been seeing claims like this for years and every time it's been total bullshit. 99.9% chance it is this time as well, but enjoy the thought experiment.
And yet we have somehow gone from rechargeable phone batteries that were about 3 times bigger than the phone I'm typing this on and had a capacity of about 500 mAh to where we are now with the battery that powers my phone being some small part of it and having a capacity of 3000 mAh, with only two major technology changes on the way. Meanwhile, we've been using the same technology for over a decade and the capability keeps getting better. I wonder why that is?
Sometimes it's not pure bullshit, but instead intentionally misses details
Like articles going "new battery lasts 1000 years in one charge!" - which is true of Nuclear Batteries, because they give basically a maximum of 1 watt of energy per hour. (Which is useful for very specific purposes like a pacemaker)
Are you saying Grandma’s a WMD?
Careful, 'Murica is gonna invade your grandma to bring democracy to her organs.
The problem is that batteries must meet a whole set of other criteria as well to be competitive, for example cost and energy density. If they are not mentioned, they are probably worse in that aspect. Which just means they are still useful for some applications, just maybe not for cars, laptops or cellphones.
However, this technology does not yet match the energy density of lithium-ion batteries.
It would be good if you actually told us what that energy density is...
Technically, a copper wire is a battery that charges in (a very tiny fraction of ) seconds.
Two important parts of a battery are how much energy it can store in a certain space and how much it weighs. If it is bigger and holds the same amount of energy that might be ok for a non mobile storage if it costs less, like a house. If it weighs more for a certain energy that wouldn't be useful for cars and mobile things but might be ok for small things where the weight is negligible anyway. For cars you want a small energy dense battery that is light as possible
The technology uses nickel and iron clusters smaller than 5 nanometers, meaning 10,000 to 20,000 clusters could fit within the width of a human hair.
By using these dimensions, the researchers increased the electrode surface area, allowing almost every atom to participate in the chemical reaction. This efficiency enables the battery to reach a full charge in seconds rather than the seven hours required by historical versions of the technology.
5nm nano fabrication will cost a fortune. this week's cure-all battery.
Nano chemistry is entirely different from nano fabrication. I haven't read the paper but most materials like this are made by mixing chemicals in a beaker and/or heating them in a furnace.
Yeah, that's exactly what they do. You can click through to the original article and then the paper abstract if you want, but yeah they mix graphene and protein and heat it.
Meanwhile my UPS taks 8 hours to charge and lasts 8 minutes.
UPS batteries are something i don't understand either. Why have they not changed with all the new tech we have now? Is it just still made of the best chemicals for their use and to then be recycled or something?
UPS batteries need to be fully charged all the time. Lead acid batteries like to be fully charged. Lithium batteries need to be stored around 50% charge to have a long lifetime.
Lead batteries are also cheap.
And mine take ~30 minutes to charge. This person may want to replace their batteries.
Charge time depends on the UPS. The cheap consumer grade ones usually have a float charger that takes forever.
There are newer LFP portable batteries with <10ms UPS switch times that charge quickly and will keep the power on longer. They also have much longer battery life's (3000+ cycles) , and LFP cells don't degrade the same when kept at 100% like other types, although you should still cycle them a few times a year.
Bluetti makes some, the elite series has their latest UPS features. The non elite are slower and noisier.
Its all fairly new and have been improving year over year. For example, earlier models may not have switched back on if power was out for a long time and it fully drained the battery. Now some models can turn back on.
Edit: more details.
If it lasts 30 years, it will not fly with the industry and the concept of planned obsolescence.
Ooh, they'll figure a way to make it clock out on the last monthly payment. One little chip will do, or just a few lines of code in the right place.
Someone will find a way to make it a subscription service that stops working when a certain MW is exceeded
We are heading for a subscription LIFE.
Did you ever see the movie THX 1138 (1971)?
The police stop chasing him when his "value to society" runs out.
Then a new player will become dominant in the industry.
Is it also a room-temperature superconductor and a dessert topping?
After 12,000 cycles it breaks down into rainbow sprinkles
Well maybe this time the new battery tech can be real and gay!
So a 3 megawatt charger can charge 50 kWh in one minute. That's some serious power.
And probably not at all practical.
Eeehhhhh — yeah
Aerogel. So not gonna be good for mobile applications— cars etc.
But might be workable for static applications????
Just make one large enough to power my house for 2 weeks and let me use solar completely detached from the grid. I'll put it on the side of my house.
the device achieves an excellent specific energy (47 W h kg−1) and superior specific power (18 kW kg−1)
I'm not familiar with this stuff. How does that compare to popular lithium batteries?
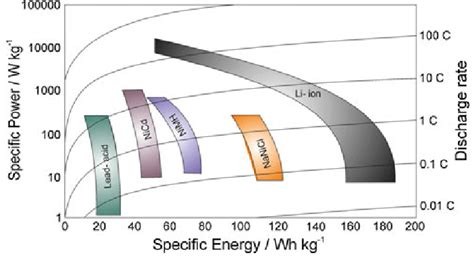
Looks like it's more like NiMH than LiPo, but higher power than NiMH (which I guess lines up with their claims of charging super fast).
Poorly. According to a random Wikipedia query, commodity lithium ion is ~270 Wh per kilogram. So this is around 20% of that, according to the above.
"Excellent" may be in comparison to other byzantine specialty battery chemistries, but lithium ion remains resolutely enthroned.
It might be cool for storing solar energy for your home, though. We don't need to always carry the battery in every use case
Nickel iron is typically used for off grid solar energy storage. Weight doesn't matter at all since the battery won't be moved. The most important thing is lifetime. Traditional nickel iron batteries last for decades and can be refurbished.
Most li-ions land around 120-160 W-h /kg. So much poorer, but much cheaper on density
The specific power (power density) is kind of crazy though. I think most li-ions top out around 10kW/kg, any more and they will overheat and boil their electrolyte which usually leads to fire.
Call me pessimistic but I'm guessing this is only time we'll be hearing about it
NIckel Iron is fantastic without any revolutionary improvements. Batteries made 100 years ago still work today. They are large and heavy so are only of use for home power.
The big "down side" which is the reason it isn't commercially developed at large scale is that they last forever. No investors are going to give billions to a business that can't generate revenue forever with a product that needs replacing every 3 years.
Abstract
Downsizing metal nanoparticles into nanoclusters and single atoms represents a transformative approach to maximizing atom utilization efficiency for energy applications. Herein, a bovine serum albumin-templated synthetic strategy is developed to fabricate iron and nickel nanoclusters, which are subsequently hydrothermally composited with graphene oxide. Through KOH-catalyzed pyrolysis, the downsized metal nanoclusters and single atoms are embedded in a hierarchically porous protein/graphene-derived carbonaceous aerogel framework. The carbon-supported Fe subnanoclusters (FeSNC) as the negative electrode and Ni subnanoclusters (NiSNC) as the positive electrode exhibit remarkable specific capacitance (capacity) values of 373 F g−1 (93 mAh g−1) and 1125 F g−1 (101 mAh g−1) at 1.0 A g−1, respectively. Assembled into a supercapacitor-battery hybrid configuration, the device achieves an excellent specific energy (47 W h kg−1) and superior specific power (18 kW kg−1), while maintaining outstanding cycling stability of over 12 000 cycles. Moreover, FeSNCs displayed a significantly reduced oxygen evolution overpotential (η10 = 270 mV), outperforming the RuO2 benchmark (η10 = 328 mV). Molecular dynamics simulations, coupled with density functional theory calculations, offer insights into the dynamic behavior and electronic properties of these materials. This work underscores the immense potential of metallic subnanoclusters for advancing next-generation energy storage and conversion technologies.
Herein, a bovine serum albumin-templated synthetic strategy is developed to fabricate iron and nickel nanoclusters, which are subsequently hydrothermally composited with graphene oxide.
Is this how Doom starts?